
Agglomeration : Drum agglomerators
What is the agglomeration process ?
What causes agglomeration of particles ?
Follow us on Twitter ![]()
Question, remark ? Contact us at powder.process@protonmail.com
| Section summary |
|---|
| 1. What is the
agglomeration of powder particles ? |
| 2. Agglomeration
binding mechanisms |
Agglomeration processes are ubiquitous in the process industry, be it wanted agglomeration through a process, or unwanted agglomeration like for example caking. This page is aiming at explaining what are the physical phenomena that cause agglomeration of powder particles, as such an understanding is critical to ensure that a factory is operating properly such processes as fluidized beds agglomerators, roller compactors, tablet presses, extrusion or sintering.
1. What is the agglomeration of powder particles ?
The agglomeration is the action to gather originally separated solids particles into a conglomerate, which is a mass of particles which adhere against each other.
The agglomeration process can be carried out thanks to different agglomeration technologies, the most common are the following :
- Disc agglomerators
- High shear mixes
- Tumblers
- Fluidized bed agglomerators
- Tablet presses
- Roller compactors
- Extrusion
- Sintering
Each of this technology is actually playing on a different mechanism to ensure that solid particles form an agglomerate solid enough to meet the target product properties.
2. Agglomeration binding mechanisms
The agglomeration of solids particles can be due to 5 different categories of binding mechanisms, for a specific agglomeration process, one or several of these mechanisms can be at play.
2.1 Solid bridges
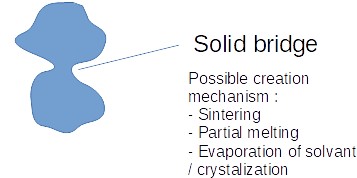
Solid bridges are the stronger link that can be made in between particles during agglomeration as they become directly linked by solid.
Solid bridges can be created by the following phenomena :
- Sintering (the particles, sufficiently heated up, are "merging")
- Partial melting of the contact surfaced in between particles (different than the sintering principle described above)
- Chemical reactions
- Recrystalization, which often happens when water is present, dissolves the solid under certain conditions, and is then evaporated. It can also be done by using a solution with some colloidal material used to wet the particles of solids and which is afterwards evaporated leaving the colloidal particles create solid bridges.
2.2 Adhesion and cohesion forces

Those forces happen typically when a layer of a certain substance is in between 2 particles.
It is the mechanism of agglomeration used by viscous liquid binders which are are coming in between the particles and stick them together. The binder is added during the agglomeration process, depending on the quantity added, it can even turn into a matrix binder, which means that the whole space in between particles, not only a layer, is occupied by the binder.
The particles can also adsorb molecules at their surface. This very thin layer, for rather small particles, can play a role as 2 adsorption layers on 2 particles can attract and keep together the 2 hosts particles.
2.3 Surface tension and capillary forces : liquid bridges
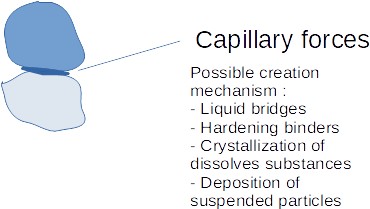
The presence of a liquid, most often water, is one of the major causes of agglomeration of particles. Water at the surface of the solids particles can create liquid bridges thanks to free water, or capillary condensation. When water is filling the pores of the solids until the surface, a negative capillary pressure within the pore can develop, creating a force at the end of the liquid bridge and keeping the particles together.
Those liquid bridges happen at the "touch point" or coordination point in between 2 particles.
2.4 Attraction forces between solids
Those forces are significant at very small scale, in between molecules, at can be high at this scale, but as soon as the distance in between particles increases, those forces become negligible compared to the other mechanisms mentioned in this page. As they act at very close range in between particles, they are also mainly significant for very fine particles (few dozens microns, or even nano particles).
Top
5 Most Popular
1.
Pneumatic transport design guide
2. Ribbon
blenders
3. Powder mixing
4. Hoppers design guide
5. Measuring degree of
mixing
--------------
 --------------
--------------
Top 5 New
1.
Continuous Dry Mixing
2. Mixing speed
3. Mixer cycle time optimization
4. Batch / continuous mixing
comparison
5. Energy Savings
Attraction forces can be of molecular, electrical or magnetic origin. The forces involved are the following :
- Van der Waals forces
- Valence forces
- Non valence associations (hydrogen links for example)
- Electrostatic forces
- Magnetic forces
2.5 Interlocking

The interlocking phenomena, helping particles to stay together in a conglomerate, is related to the shape of the particles and how the mechanically block the movement of each other at the particle level. For example, fibers, long particles can play a role to interlock with some other particles and restrain their movement. It is also the case in compression of powder where some particles can break, deform, and "enroll" around other particles, strengthening the bond in between them.
Source